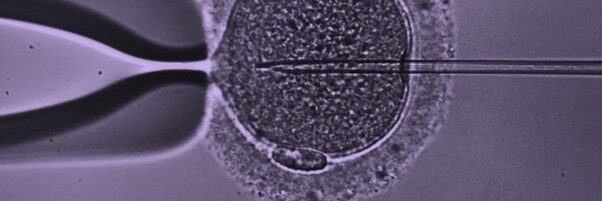
Welcome to the Leeds Centre for Reproductive Medicine (LCRM) website.
How to contact us
-
- Seacroft Hospital
York Road
Leeds
LS14 6UH -
- Our admin team are available Monday to Friday 8am to 5pm.